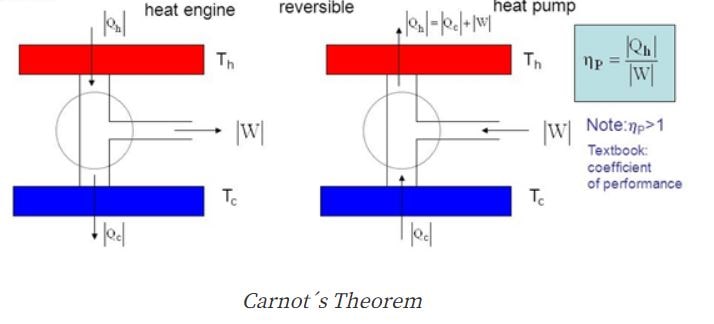
What is Carnot cycle?
The Carnot cycle is the ideal type of thermodynamic cycle. We got proof from the Carnot theorem that the Carnot cycle offers an upper limit on the efficiency of any thermodynamic engine during the conversion of heat into work. In a Carnot cycle, the engine transfer energy in the form of heat between two thermal reservoirs at the known temperatures TH and TC, and this transferred energy is converted to the work done by the system. On the other hand, to create the temperature difference through the application of work to the system the efficiency of the refrigeration system is used. However, Carnot engine efficiency is an important thing; have a look at Carnot engine efficiency formula derivation.
What is the efficiency of Carnot engine?
The Carnot engine is the cycle of theoretical thermodynamics which has been invented by Nicholas Leonard Sadi Carnot, who made this Carnot Engine with a basic model in 1824. This engine also known as the theoretical engine handles the Carnot Cycle. It approximates the highest efficiency of a heat engine while it was in the process of conversion and it was functioning within the two reservoirs which can process successfully.
Following the internal energy of the gas might be the same within the resolution of the cycle,
W= Q_H – Q_C
Here, is a fraction of the heat contributed into the hot reservoir ahead of the red isotherm.
is the heat that has been discarded when the gas is flattened ahead of the cold isotherm.
The fraction of the heat input that has been transformed to work properly can be recognized as efficiency.
Efficiency = W/Q_H
= (Q_(H- ) Q_c)/Q_H
To experience better efficiency it is important to improve the concept of entropy.
W = Q_H – Q_C
= (1- T_C/T_H ) Q_H
As a result, the efficiency of the engine is the fraction of the accessible heat energy that can be transformed to work
Efficiency = W/Q_H
= 1- T_C/T_H
The temperatures are considered as the degrees Kelvin. For instance, the efficiency of the Carnot efficiency is within 1- (273/373) = 0.27 then the engine might be having hot reservoir within the boiling water and a cold reservoir within the ice-cold water, and then the habitat of the heat energy can be transfigured into the applicable work. The attempt to build a better heat engine reduces the losses made by friction. Sometimes, it might be very difficult to identify the 27% efficiency while intervening within 0 C to 100 C.
In the 18th century, stream locomotives were invented and the weight or power ratio which can be helpful to move forward within a track obtained by carefully utilizing high-pressure boilers, which contain the boiling water at adequate atmosphere pressure. Whenever the atmospheric pressure is within 6, the boiling temperature might be within 280 C. In this situation, the system might be working within the 300K room temperature which provides the conceptual efficiency within 250/550 or within 45% which can be considered a great advancement.
Carnot engine efficiency formula derivation
Carnot engine efficiency formula derivation is efficiency = WQH=1-TCTH. The Carnot Cycle creates a better engine. The diagram of P-V has outlined the performance of the Carnot Engine, in here, the “working fluid” that lengthens and shrinks inside the cylinder which is filled with gas. It is noted that you may see solar panel efficiency calculations.
The high temperature has been considered as the T1 and the low temperature can be recognized as the T2 sustained as the ideal gas thermometer, which is
T = P^V/NR
The cycle of the Carnot Engine performs as follows:
1. Leg 1: Isothermal Expansion within the maximum temperature. It elevates a big bundle of sand when the gas lengthens, which is indicating that the │W1│ not working. The expansion might keep the temperature low. To maintain this temperature, the gas should soak the nearby heat │Q1│.
2. Leg 2: Adiabatic Expansion. As the gas lengthens, it elevates the large stockpile of sand; it does perform better at │W2 │. The expansion keeps the temperature low. Although, it is adiabatic no heat can be soaked.
3. Leg 3: Isothermal Expansion within its low temperature. When the gas began to deal with the small stockpile of sand which refers that the │W3│has been completed. Naturally, the compression dispatches the temperature toward the top. To maintain this temperature, the gas should reduce the nearby excess heat │Q2│.
4. Leg 4: Adiabatic Compression. When the gas begins to deal, it simply authorizes the small stockpile of sand downward; the work │W4│ has been completed within the gas. Since its Adiabatic, no heat has been soaked.
How to calculate the Carnot engine efficiency?
To calculate the efficiency of the Carnot Engine, two temperatures are needed which can also be recognized as the two reservoir works within the system. One is the cold reservoir which will be referred to as the temperature Tc and the hot reservoir will be referred to as the. The efficiency of the Carnot is connected with the greatest amount of work that can be accomplished with the differences made within the temperature.
n = (TH – Tc/ TH )×100%
0≤n≤100 %
For instance,
T_C= 300 K (~27℃)
T_H= 400 K ( ~ 127℃)
n = (1- T_C/T_H ) ×100 %
= (1 – 300/400 ) ×100%
Why Carnot engine is most efficient?
The Carnot is improvable delineating the highest limit of the efficiency for the engine cycle. Some experimental engine cycles are irresistible and they usually have the lowest temperature than the Carnot Cycle which operates within the same temperature. There is a factor that regulates the efficiency performed as the addition of the working fluid and its abolition. The Carnot Cycle receives the highest efficiency since; every heat is connected with the working fluid within the highest temperature. For this situation, the working fluid performs as the ideal gas for the Carnot’s engine.
There are some principles of the Carnot Engine and these principles are made for the cyclical devices, for instance, the heat engines, which include:
1. The efficiency of the absolute heat engine may always remain below the efficiency of the one which is reversible that has been performing better which is connecting the two reservoirs.
2. The efficiency of the reversible heat engines is performing better within the same two reservoirs and they both are considered identical.
To improve the thermal efficiency of the gas power turbine, it is truly important to enhance the temperature of the burning chamber. Some blades of the turbine may not tolerate the maximum temperature of the gas and guide toward exhaustion.
To improve the efficiency of the Carnot Engine some tasks can be completed,
The efficiency of the Carnot engine:
n = 1- (T2/T1)
T1 and T2 are considered as the temperature of the sink and the source. The efficiency of the engine can be maximized up to 100% and if the T2 = 0 can be maintained as the temperature of the sink.
Final thought
We know that efficiency is a major part of all devices. If we divided output by input and multiply by 100 then w will get the general efficiency. In a general sense, how much work is done by an engine within a specific time? It means how much energy we spend and how much work is done by it which is called Carnot engine efficiency. However, we have referred to the Carnot engine efficiency formula derivation and it may be a sufficient description of Carnot engine efficiency. Stay with openread and see the related topic.
What is Binary Cycle Geothermal Plant?

